2. Oeffinger KC, Mertens AC, Sklar CA, et al. Chronic Health Conditions in Adult Survivors of Childhood Cancer. N Engl J Med 2006; 355(15):1572–1582.


6. Maron BJ, Chaitman BR, Ackerman MJ, et al. Recommendations for Physical Activity and Recreational Sports Participation for Young Patients With Genetic Cardiovascular Diseases. Circulation 2004; 109(22):2807–2816.


7. Reineck E, Rolston B, Bragg-Gresham JL, et al. Physical activity and other health behaviors in adults with hypertrophic cardiomyopathy. Am J Cardiol 2013; 111(7):1034–1039.


8. Coats CJ, Rantell K, Bartnik A, et al. Cardiopulmonary Exercise Testing and Prognosis in Hypertrophic Cardiomyopathy. Circ Heart Fail 2015; 8(6):1022–1031.


9. Ingles J, Yeates L, Hunt L, et al. Health status of cardiac genetic disease patients and their at-risk relatives. Int J Cardiol 2013; 165(3):448–453.


10. Olivotto I, Maron BJ, Tomberli B, et al. Obesity and its Association to Phenotype and Clinical Course in Hypertrophic Cardiomyopathy. J Am Coll Cardiol 2013; 62(5):449–457.


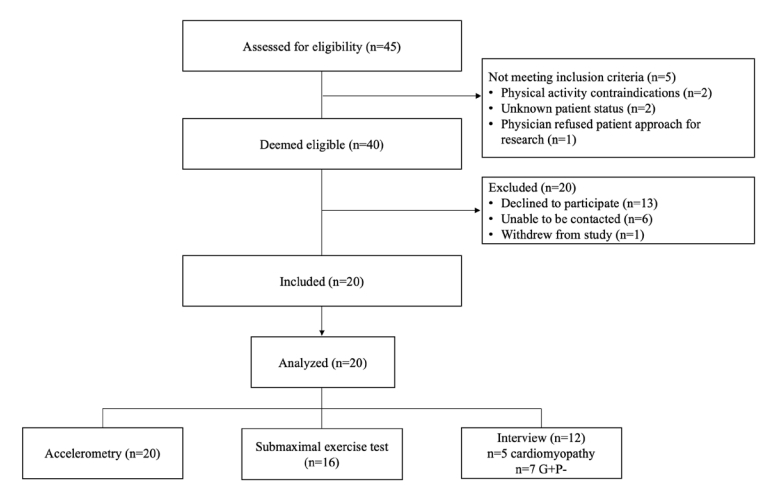
12. Leech NA, Onwuegbuzie A. Guidelines for Conducting and Reporting Mixed Research in the Field of Counseling and Beyond. J Couns Dev 2010; 88(1):61–69.

13. Braun V, Clarke V. Using thematic analysis in psychology. Qual Res Psychol 2006; 3(2):77–101.

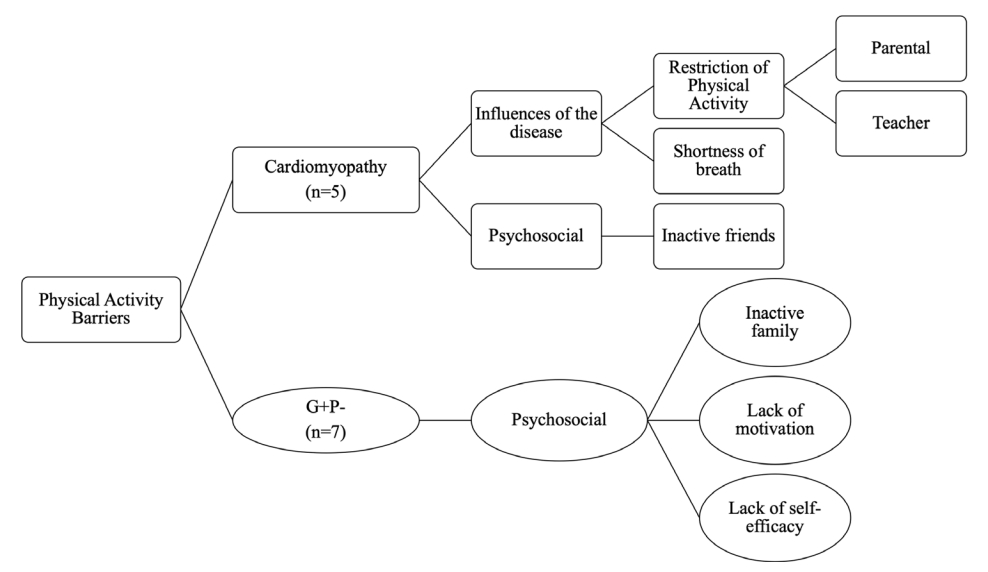
14. Moola F, Fusco C, Kirsh JA. What I Wish You Knew. Social Barriers Toward Physical Activity in Youth With Congenital Heart Disease. Adapt Phys Activ Q 2011; 28:56–77.

15. Trost SG, Pate RR, Freedson PS, Sallis JF, Taylor WC. Using objective physical activity measures with youth: How many days of monitoring are needed? Med Sci Sports Exerc 2000; 426–431.


16. Puyau MR, Adolph AL, Vorha FA, Zakeri I, Butte NF. Prediction of Activity Energy Expenditure Using Accelerometers in Children. Med Sci Sports Exerc 2004; 1625–1631.

17. Colley RC, Garriguet D, Janssen I, Craig CL, Clarke J, Tremblay MS. Physical activity of Canadian adults: accelerometer results from the 2007 to 2009 Canadian Health Measures Survey. Health Rep 2011; 22(1):7–14.
18. Cumming GR, Everatt D, Hastman L. Bruce treadmill test in children: normal values in a clinic population. Am J Cardiol 1978; 41(1):69–75.


19. Rhodes J, Ubeda Tikkanen A, Jenkins KJ. Exercise Testing and Training in Children With Congenital Heart Disease. Circulation 2010; 122(19):1957–1967.


20. Borg GAV. Psychophysical bases of perceived exertion. Med Sci Sports Exerc 1982; 14(5):377–381.


21. Schofield C. An annotated bibliography of source material for basal metabolic rate data. Hum Nutr Clin Nutr 1985; 39(S)1:42–91.
22. Tremblay MS, Carson V, Chaput JP, et al. Canadian 24-Hour Movement Guidelines for Children and Youth: An Integration of Physical Activity, Sedentary Behaviour, and Sleep. Appl Physiol Nutr Metab 2016; 41(6 Suppl 3):S311–327.

23. Colley RC, Carson V, Garriguet D, Janssen I, Roberts KC, Tremblay MS. Physical activity of Canadian children and youth, 2007 to 2015. Health Rep 2017; 28(10):8–16.

24. Moola F, Faulkner GE, Kirsh JA, Kilburn J. Physical activity and sport participation in youth with congenital heart disease: perceptions of children and parents. Adapt Phys Activ Q 2008; 25(1):49–70.


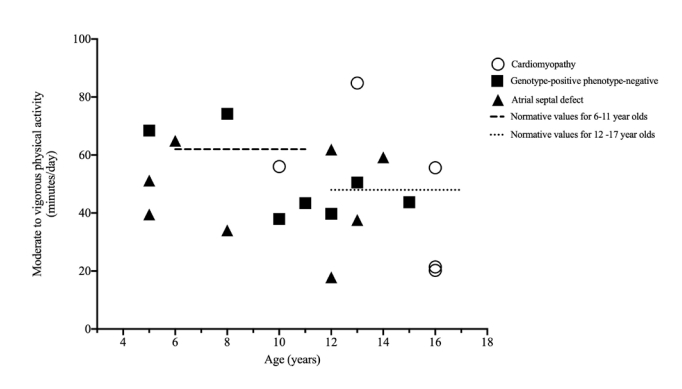
26. Voss C, Duncombe SL, Dean PH, de Souza AM, Harris KC. Physical Activity and Sedentary Behavior in Children With Congenital Heart Disease. J Am Heart Assoc 2017; 6(3):

28. Allison KR, Dwyer JJ, Makin S. Perceived barriers to physical activity among high school students. Prev Med 1999; 28(6):608–615.


29. Gunnell KE, Brunet J, Wing EK, Bélanger M. Measuring Perceived Barriers to Physical Activity in Adolescents. Pediatr Exerc Sci 2015; 27(2):252–261.


31. Massin MM, Hövels-Gürich HH, Gérard P, Seghaye MC. Physical activity patterns of children after neonatal arterial switch operation. Ann Thorac Surg 2006; 81(2):665–670.


33. McManus A, Leung M. Maximising the clinical use of exercise gaseous exchange testing in children with repaired cyanotic congenital heart defects: the development of an appropriate test strategy. Sports Med 2000; 29(4):229–244.

34. Allor KM, Pivarnik JM. Use of Heart Rate Cutpoints to Assess Physical Activity Intensity in Sixth-Grade Girls. Pediatric Exercise Science 2000; 12:284

35. Butte NF, Watson KB, Ridley K, et al. A Youth Compendium of Physical Activities: Activity Codes and Metabolic Intensities. Med Sci Sports Exerc 2018; 50(2):246–256.

36. Longmuir PE, Brothers JA, De Ferranti SD, et al. Promotion of Physical Activity for Children and Adults With Congenital Heart Disease. Circulation 2013; 127(21):2147–2159.