Statins Do Not Impair Whole-body Fat Oxidation During Moderate-intensity Exercise in Dyslipidemic Adults
Article information
Abstract
Objectives
Some lipid-lowering agents, for example, nicotinic acid and fibrates, decrease an individual’s ability to oxidise fat during exercise. However, it is unclear whether statins affect whole-body fat oxidation during exercise in patients. This study investigated whether fatty acid oxidation is impaired in a dyslipidemic population, while walking at a moderate intensity.
Methods
Patients (n=16), walked for 45 minutes on a treadmill at 50% of their estimated VO2max, in the absence and presence of their prescribed statin. Fat oxidation was investigated by examining respiratory data, and circulating plasma glycerol and free fatty acids.
Results
Analysis of respiratory data indicated a progressive increase in fat oxidation over time, along with a decrease in carbohydrate oxidation, for all patients during exercise, in both the absence and presence of a statin (P≤0.05). The increase in the percent of energy derived from fat was further supported by the observation of a significantly progressive increase in circulating glycerol and free fatty acids during the exercise period. However no significant difference in the extent of change was observed when comparing the respiratory and biochemical response to physical activity in the absence and presence of the prescribed statin.
Conclusions
There is no evidence of a negative impact of statins on the ability to use fat as a fuel for moderate-intensity aerobic exercise. Given the importance of physical activity, this result encourages patients to exercise by walking regularly, with the confidence that substrate metabolism is unaltered in the presence of this class of lipid-lowering drug.
INTRODUCTION
Lipid-lowering drugs are amongst the most commonly subsidized prescribed drugs world-wide, including Europe and the United States (USA). In 2016, atorvastatin and simvastatin were ranked in positions 1 and 7 in the USA [1]. In the same year, in Australia, atorvastatin and rosuvastatin were in positions 1 and 3, with the number of Australians per thousand taking the drug daily being 53 for atorvastatin and 34 for rosuvastatin [2].
Physiologically, these drugs inhibit liver enzyme HMG-CoA reductase, blocking cholesterol synthesis. They are recognized as effective in the secondary prevention of cardiovascular disease and all-cause mortality [3]. However, it is now well established that a healthy diet and regular physical activity are valuable contributors to maintaining healthy blood lipids, with exercise improving cardiovascular fitness; reducing all-cause mortality and the risk of coronary heart and cardiovascular disease, stroke, type 2 diabetes mellitus (T2DM), and obesity. A recent meta-analysis highlighted that physical activity alone is potentially as effective as many drug interventions on mortality outcomes in coronary heart disease, stroke, heart failure, and prediabetes [4]. Despite this, there exists a bias against exercise intervention with a subsequent progression towards lowering the threshold for pharmaceutical treatment, and an increased likelihood that individuals embarking on an exercise regime are already taking a cholesterol-lowering agent as a secondary or even primary prevention against cardiovascular risk. To maximize the benefit of pharmaceutical intervention and exercise, it is important therefore, to understand the effect of these drugs on the body’s response to exercise.
Lipid stores, critical for fueling prolonged exercise, exist as triglycerides in adipose tissue and muscle, and also as circulating triglycerides. Hormone-sensitive lipase (HSL), an intracellular enzyme in adipose tissue, and lipoprotein lipase (LPL), an enzyme which hydrolyzes the triglyceride core of circulating chylomicrons, both liberate free fatty acids (FFA) for use by exercising muscle. Any drugs acting at these sites, could potentially alter FFA availability, which may result in increasing dependence on muscle glycogen stores and possible earlier fatigue [5]. An early study examining muscle biopsies of quadriceps femoris has documented glycogen depletion in healthy males while cycling in the presence of the lipid-lowering drug acipimox, also known as nicotinic acid [6]. A detailed review has highlighted that pharmacological doses of acipimox have a greater adverse effect on exercise performance in individuals whose carbohydrate stores are already limited [7]. In addition, another study has shown that prolonged cycling performance in the presence of acipimox is improved if carbohydrate is regularly supplemented during the exercise session [8]. Other studies in healthy volunteers undertaking exercise have found in addition to acipimox, that the lipid-lowering fibrates such as bezafibrate and gemfibrozil, also have a negative impact on fat oxidation [5,9]. However, no negative impact of simvastatin, fluvastatin or atorvastatin has been observed in healthy exercising participants [5,9,10].
In contrast, the impact of statins on fat metabolism in patient cohorts is less clear. The available research in patients undergoing exercise has produced conflicting results, with studies confounded by the co-administration of a kaleidoscope of other cholesterol-lowering agents, or with β-blockers, which inhibit the release of FFA from adipose tissue and complicate the interpretation of the data [11,12]. One study in patients has reported reduced fat oxidation with atorvastatin, during lower-intensity exercise [13]. Patients have also self-reported increased fatigue with exertion after taking simvastatin for a period of 6 months [14]. In contrast, other studies, including a recent review, have reported no negative impact of statins on fat metabolism [15,16].
Given the importance of exercise in these patients, and the limited and conflicting data on the interaction between cholesterol-lowering agents and exercise, the current study investigated in a dyslipidemic population, whether statins impair whole-body fat oxidation during exercise of moderate-intensity. This study is unique as it appears to be the first investigation of whole-body fat oxidation in both the absence and presence of a statin during exercise, within the same group of patients.
METHODS
Participants
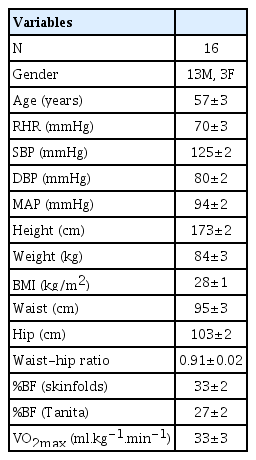
The study engaged 16 Caucasian adults taking a statin for hypercholesterolemia (Table 1). The study was approved by the Human Research Ethics Committees from The University of New South Wales (UNSW) (HREC05310) and the Sydney Adventist Hospital (HREC12/06) and all procedures were in accordance with institutional guidelines. Participants were recruited by newspaper advertisements, and required written permission from their medical practitioner to participate. Individuals also provided written informed consent, and were aware of their right to withdraw at any time. Participants were considered able to engage in physical activity comprising of a brisk walk, by satisfactorily completing a physical activity readiness questionnaire (PAR-Q), as well as an additional questionnaire on family history, medical history, present symptoms, and current pattern of physical activity [17,18]. Patients with T2DM were excluded as this condition may influence whole-body fat oxidation. In addition, recruited participants were not taking other medications which could knowingly interfere with fat oxidation.
Experimental design
Each participant made a total of three visits to the laboratory. All participants ceased lipid-lowering medication for a minimum of three weeks prior to Visit 1. The purpose of Visit 1 was to (a) prescreen the participant, (b) perform a submaximal exercise test on a treadmill to predict the participant’s maximum oxygen uptake (VO2max), and (c) calculate the individual treadmill settings (speed and gradient) which would enable the participant to walk at a power output of 50% VO2max in Visits 2 and 3. The latter were identical to each other with the notable exception that Visit 2 continued in the absence of the drug and Visit 3 in the presence of the drug. Visit 3 occurred after the participant had resumed their lipid-lowering medication for a minimum of 3 weeks. Statin administration was verified verbally and by assessment of plasma lipids measured at the end of the drug versus no drug phases, on the mornings of Visits 2 and 3.
Visit 1: Prescreening and submaximal exercise test
Resting heart rate (HR), blood pressure, anthropometric assessment (height, weight, body mass index (BMI), waist and hip circumference), and percent body fat by bioelectrical impedance (Tanita BWB-800, Tanita Corporation, Japan) and by skinfolds analysis (biceps, tripceps, subscapular, suprailiac and abdominal) was measured [19].
Participants performed a submaximal exercise test on a treadmill to predict VO2max and to calculate the individual treadmill setting for each participant to exercise at 50% of their maximal capability in Visits 2 and 3. Ventilation (VE, l.min-1), oxygen uptake (VO2, l.min-1), expired carbon dioxide (VCO2, l.min-1) and respiratory exchange ratio (RER) (VCO2/VO2) were measured using a metabolic cart (ParvoMedics TrueMax 2400). HR was monitored via an electrocardiogram (ECG) and a HR monitor. Participants chose a comfortable walking speed at 0% gradient. With the speed constant, exercise intensity was increased a further two times, by increasing the gradient of the treadmill belt every 3-5 min after a stable exercise HR was recorded. The predicted maximum oxygen consumption was calculated by plotting exercise HR against oxygen consumption, with a line of best fit extrapolated to the point of each participant’s maximal predicted HR [18]. The relative perceived exertion (RPE) [18] and finger prick blood lactate, confirmed participants exercised at a submaximal level.
Visits 2 and 3: Exercise at 50% VO2max in the absence and presence of a statin
The exercise trials occurred at Visit 2 (-statin) and Visit 3 (+statin).Visit 3 occurred after participants had resumed medication for a minimum of three weeks. The two visits were otherwise identical. Participants arrived at the laboratory after an overnight fast (water permitted). A forearm vein was cannulated using a 20 gauge indwelling line (BD Insyte Autoguard, Becton Dickinson, UK). As the act of venepuncture can increase some hormone levels by more than 50% [20], a stabilization period of 30 min was allowed after the cannula was inserted into the forearm vein, and before collection of a fasting blood sample (6ml) into vacutainer tubes (Becton Dickinson, UK) containing ethylenediaminetetra- acetic acid (EDTA). Fasting blood lipids, lactate and glucose were measured immediately. Participants then consumed a low fat 1MJ CHO meal, consisting of Special K cereal and low fat dairy or lactose free milk, followed by rest in a semi-inclined position. Participants commenced exercise 90 min after their meal. They walked on the treadmill at an intensity of 50% of their calculated VO2max (determined in Visit 1), corresponding to a ‘brisk walk’ for 45 minutes. The treadmill speed for each participant was that of Visit 1, and the treadmill gradient was determined using the formula: VO2=(0.1 x S)+(1.8 x S x G)+3.5; where VO2 is gross oxygen consumption mL.kg-1.min-1 at 50% of estimated VO2max, speed (S) is in metres.min-1 and G is percent grade expressed as a fraction [18]. The predetermined treadmill settings (speed and gradient) for each individual participant remained constant throughout the exercise session. VE, VO2, VCO2, and RER were measured to determine the proportion of CHO and fat metabolism during exercise. Calculation of carbohydrate and fat oxidation was by the classical stoichiometric equations of indirect calorimetry [21]; with carbohydrate oxidation (g/min) =4.58VCO2–3.2VO2; and fat oxidation (g/min) =1.7VO2–1.7VCO2. HR and RPE were monitored. Additional blood (each 6ml) was collected into EDTA-containing tubes immediately before exercise, and at time 15, 30 and 45 minutes of exercise, for the immediate measurement of glucose and lactate. Remaining blood samples were centrifuged (4ºC, 1560g, 10min, Heraeus Megafuge 1.0R, Hanau, Germany) and plasma was stored at -86ºC prior to the analysis of glycerol and FFA.
Biochemistry
Lipids (total cholesterol (TC), triglycerides (TG), low density lipoprotein-cholesterol (LDL-C) and high density lipoprotein- cholesterol (HDL-C)) (Cholestech LDX, CA, USA), glucose (HemoCue Glucose 201, Sweden), and lactate (Lactate Pro, ARKRAY, Inc., Japan) were measured in whole blood by reflectance photometry. Glycerol was measured in plasma using two colorimetric assays for comparison (Sigma, USA; and Cayman Chemical, USA) and the concentration determined at 550nm using an Expert Plus Microplate reader (Asys Hitech, Austria) and a Versa Max Microplate reader (Molecular Devices, USA). FFA were measured in plasma by colorimetric assay (Cell Biolabs, USA) at 570nm using the Versa Max Microplate reader.
Food diary
Participants completed a 3-day food diary, to calculate total caloric intake, and percentages of macronutrient and micronutrient intake during two non-consecutive weekdays and one weekend day. Data were entered into a nutritional database (Serve Nutrition Systems, Australia).
Statistical Analysis
Power calculations [22], based on a previous study [5] where participants walked at 50% VO2max in the presence or absence of the lipid-lowering agent, acipimox, determined that 11 participants would be sufficient to detect a significant decrease in plasma glycerol, with an effect size of 2.17 and assuming a 0.05 significance level and 80% power. The current study recruited 16 participants. All data are expressed as mean±standard error of the mean (SEM). The Student’s paired t-test compared fasting blood lipids±statin and area under the curve (AUC) for glycerol and FFA. A two-way repeated measures ANOVA examined differences between the two within-subject factors (treatment and time) for the physiological variables (HR, RPE, fat and CHO oxidation, % energy derived from fat and CHO, RER, VO2, glucose, lactate, glycerol, and FFA), measured during exercise. Where a significant two-way interaction was found (FFA), simple main effects examining differences between trials at each level of time and treatment were determined using the General Linear Model (GLM) Repeated Measures procedure. For cases without a significant two-way interaction, the main effect for treatment and time were determined. Variables which did not meet Mauchly’s test of sphericity were interpreted using a Greenhouse-Geisser correction. For all data comparisons, unless indicated, significance is at P≤0.05. In using the GLM Repeated measures procedure for the FFA data, a Bonferroni adjustment was applied for the testing of multiple main effects, with a significance of P≤0.05/3=P≤0.016 [23]. Analysis was conducted using SPSS (SPSS Inc.,USA).
RESULTS
Patient characteristics
The study engaged 13 males and 3 females, aged 39-82 years, taking a statin for hypercholesterolemia. In addition to hyperlipidemia, some patients had cardiovascular conditions (hypertension, coronary artery disease, heart murmur, aortic valve replacement, mild myocardial regurgitation, varicose veins), and arthritis. Two participants, with the permission of their medical practitioner, ceased taking metoprolol for the entire duration of the study as it interferes with fat metabolism [11].
Patients were non-smokers or previous smokers who had ceased smoking 15-35 years earlier. Body composition data revealed that participants were on average overweight, had an increased disease risk relative to weight and waist circumference, and a moderate disease risk based on waisthip ratio [18].
Regarding physical activity, participants reported an average occupational activity intensity of 1.5±0.2 (1=sedentary; 5=heavy labour), sport and leisure physical activity intensity of 6±0.4 (1=very light, 10=intense breathless), and exercise duration of 8±1 hours per week. Activities included cycling, swimming, board paddling, running, walking, dance, stair climbing, gym (circuit, weight training, aerobic classes), tennis, rollerblading, and martial arts. Aerobic fitness, indicated by a predicted VO2max, was categorized as ‘average’ for males (35±3 ml.kg-1.min-1) and ‘fair’ for females (23±0.5 ml.kg-1.min-1) [18]. The average percent daily consumption of protein and CHO was slightly higher, and lower, than the recommended 10-15% and 50-60%, respectively. The average percent daily consumption of <30% total fat was as recommended [24] (Table 2).
Lipid-lowering agents and blood lipoproteins
Of the 16 patients, 11 were taking atorvastatin, with a daily dose range of 10-80mg (1=10mg, 6=20mg, 3=40mg, 1=80mg). Rosuvastatin, at 10 mg/day was prescribed for 1 patient, and the remaining 4 patients were administering simvastatin at a dose range of 10-40mg/day (1=10mg, 1= 20mg, 2=40mg). Patients had been taking their drug for 3.5±0.6 years (range 0-10 years). There was one newly diagnosed individual who had not yet commenced his statin medication. All patients ceased taking their statin for 3.4±0.2 weeks prior to their blood lipids analysis in Visit 2. Following this visit, patients recommenced taking their medication for 26±0.7 days. A significant decrease was observed in TC, LDL-C and TC/HDL-C when taking a statin, compared with in the absence of the drug (Table 3). A trend toward a decrease in TG was also observed (P=0.073).
Energy expenditure and metabolism during exercise
All participants completed the exercise tests. The average room temperature and start times for the tests were 24±0.75 and 23±0.8 ºC; and 10.24 am ± 6 min and 10.23 am ± 8 min, respectively, in the absence and presence of the statin. Exercise performed at 50% of VO2max for 45 minutes required a total energy expenditure of 268±20 kcal or 282±21 kcal in the absence or presence of a statin.
No two-way interaction (between treatment and time) was found for fat or CHO oxidation, % energy derived from fat or CHO, RER, VO2, glycerol, glucose, lactate, HR, and RPE (Table 4). There was a two-way interaction between treatment and time for FFA (F(1.171, 12.879)=5.366, p=0.033, epsilon=0.58) at 45 minutes (P=0.024). However, this effect was disregarded following correction for the testing of multiple main effects, with a Bonferroni adjustment, (P<0.016), resulting in non-significance [23].

Physiological variable measured in participants during a walk at time = 15, 30 and 45 minutes in either the absence(-) or presence(+) of a statin.
As there was no 2x3 interaction, the main effect for treatment (irrespective of time point) and the main effect for time (irrespective of treatment) were examined. Regarding treatment, there was no difference between the conditions (±statin) for fat oxidation, carbohydrate oxidation, plasma glycerol or plasma FFA (P>0.05). The main effect of time was significant for all of these variables (P≤0.05).
To confirm no significant difference for glycerol±statin, plasma samples were analysed in duplicate, using two separate assays (Sigma and Cayman Chemical), each producing a similar result. Furthermore, an AUC analysis for glycerol (Sigma) (3753±310 and 3267±401 μmol/l, p=0.122), glycerol (Cayman Chemical) (6075±626 and 5010±373 μmol/l, p=0.106), and FFA (2608±366 and 2226±480 μmol/l, p=0.334) (each in the absence and presence of a statin, respectively), further confirmed no significant difference for the circulating plasma markers between the two test conditions.
DISCUSSION
The current study has shown no evidence of an adverse effect of statins in dyslipidemic patients on the ability to use fat as a fuel during a moderate-intensity 45-minute walk. The results in this patient group are in agreement with previous research in healthy volunteers [5], where simvastatin treatment had no impact on whole-body fat oxidation or circulating plasma free fatty acids, glycerol and glucose during walking. This result is encouraging for patients, given that the importance of regular physical activity is recognized as a positive contributor to improving and maintaining health [4]. Indeed, this study supports positive messaging to patients taking statins; that they may engage in regular exercise. This does to some extent counteract the recently suggested association of statins with an increased risk of T2DM [25]. Furthermore, implementation of a healthy diet and regular aerobic exercise, while on statin medication, may for some patients, enhance the possibility of a medically managed reduction in their statin dose or even complete cessation of this medication. The current study provides reassurance for both patients and their advising physicians, that it is possible to engage effectively in moderate-intensity exercise without the prescribed statin compromising the ability to use fat stores as a fuel to carry out the exercise session.
The lack of an adverse effect on exercise-induced fat oxidation by statins may be due to the fact that their action is thought to be largely confined to the liver [26], rather than adipose tissue. Preliminary research by our group on ezetimibe (Ezetrol), a non-statin whose action is confined to inhibiting cholesterol absorption from the intestine [27], has also shown no effect of this drug on whole-body fat oxidation during 45 minutes of moderate-intensity walking (data not published). In contrast, acipimox and fibrates, significantly inhibit HSL in adipose tissue, which results in inhibiting the release of FFA during exercise from this site [5,9]. Fibrates may also reduce availability of FFA to skeletal muscle, by increasing the uptake of FFA into liver with the induction of a fatty acid transporter protein [28].
The findings from this study, therefore make a valuable contribution to the somewhat confusing information on statins and metabolism. It has been suggested that statins, by lowering cholesterol, may affect mouse membrane calveolae, important in fatty acid transport [29]. Statins have been implicated in impairing mitochondrial function, evidenced by increased lactate production, elevated intramuscular lipid stores, decreased mitochondrial activity (measured by a decrease in cytochrome C activity) and lower CoQ10 levels [30,31]. However, despite the above, it has been found that a decrease in circulating CoQ10 does not result in a reduction in muscle CoQ10 [32]. Furthermore, reviews to date, have not concluded that CoQ10 is required when taking statins [33,34].
Other conflicting results include reports in humans of increased resting RER with statins, indicating a possible shift from fat to CHO metabolism [13,35]. A 12-week aerobic training study measured a greater increase in VO2max in a -statin, compared with +statin group. The authors reported a decline in muscle citrate enzyme activity, suggesting that statins may affect mitochondria [36]. However, in contrast to the above, there are reports of simvastatin therapy in patients having no effect on RER or aerobic fitness (VO2max) suggesting no effect on fat metabolism and aerobic capacity [34]. Additionally, a 30% increase in VO2max, indicating an increase in aerobic fitness, has been reported in patients on rosuvastatin undergoing exercise training for 10 weeks [16].
The conflicting results in the literature, are due to many variables, including that of studying different patient groups in either the presence or absence of the drug. The current study eliminated this variable, adopting a cross-over design, with all participants required to undertake exercise with and without a statin. Based on previous research in healthy volunteers [5], the current study adopted a 3-week treatment period verified by assessment of plasma lipids at the end of the drug versus no drug phases. The treatment period significantly lowered TC and LDL-C, confirming that it was of sufficient length to obtain the desired effect of lowering circulating lipids, enabling patients to serve as suitable controls.
To minimize disruption to patients, the current study adopted a non randomized cross-over design, with all patients experiencing the prescreening session and the first 45-minute walk in the absence of the drug, and the second 45-minute walk in the presence of the drug. Additionally, researchers did not interfere with each individual patient’s statin type and therapeutic dosage, to keep with their real-life situation, controlled by their physician. This, along with the age range of the patients, was not considered a limitation, as the strength of this study is in having each patient serve as their own effective control.
This study also concentrated on the effect of statins without the confounding variable of decreased blood glucose during exercise. The mean energy expenditure during exercise was 1.1MJ (282 kcal), well below the CHO energy reserve of 8-11MJ for a well-nourished adult [5,37]. Additionally, participants consumed a low fat 1MJ CHO meal 90 minutes prior to exercise. Blood glucose levels remained stable during both trials. The decreasing contribution of CHO and increasing contribution of fat as a fuel during the walk was similar in the ±statin exercise sessions. At 45 minutes of walking, fat oxidation peaked at an average of 0.28g/min.
Fat and carbohydrate oxidation were measured in the current study by indirect calorimetry, which is considered to be highly reliable for the determination of substrate use during exercise. Plasma FFA is considered to be a somewhat imperfect indicator of FFA kinetics as it is taken up by muscle, splanchnic and other tissues during exercise [38,39]. Plasma glycerol is considered more reliable than plasma FFA as a measure of lipolysis, however it too can be taken up by skeletal muscle, and the assumption that plasma glycerol is a measure of total FFA released into plasma needs to be treated with caution [40]. In the current study, analysis of plasma FFA and glycerol showed individual variability, with some cases increasing and then decreasing in the levels of these markers, with an overall group average increase in FFA and glycerol as the exercise progressed. The analysis of blood however, was selected to complement the more superior continuous sampling of respiratory gases by indirect calorimetry, with the overall finding of no adverse effect on fat oxidation by statins in the exercising participants.
The chosen modality and intensity of exercise, with a duration of 45 minutes, was adopted as it has been reported as manageable for patients and ideal for maximizing cardiovascular health benefits, significantly reducing morbidity and mortality [5,41]. Though it appears that statins do not impact fat metabolism when exercising under these conditions, future studies examining different exercise modalities, intensities and duration, are recommended.
CONCLUSIONS
There are many people world-wide on statins. Amongst this group are individuals who are considered capable of adopting a healthier lifestyle incorporating regular exercise. This study demonstrates that statins do not compromise exercise-induced fat metabolism during moderate-intensity aerobic exercise, and therefore do not contribute to premature fatigue and an inability of the patient to complete the exercise. More emphasis may therefore be placed on encouraging regular physical activity in patients to maximize the benefits of concomitant therapies such as pharmaceutical intervention.
Acknowledgements
The authors thank Associate Professor Stephen Boutcher from UNSW, for use of the metabolic cart, and for his advice and encouragement.
Notes
Funding Source
This study was funded by a grant (051101) from the Australasian Research Institute (ARI), at the Sydney Adventist Hospital; and by a Career Advancement Fund awarded to Maria Matuszek from the University of New South Wales.
Conflict of Interest
The authors declare no conflict of interest exists with respect to research, authorship and publication.
Authors’ contribution
MM and RG contributed to the conception of the study. MM designed the study, acquired, analysed and interpreted the data, and drafted the manuscript. MM and RG critically revised the manuscript. Both gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.